Biochemistry is a three-dimensional subject but most of us aren't comfortable thinking in three dimensions. For example, we often have difficulty envisaging how a three-dimensional substrate binds to a three-dimensional enzyme.
The problem is exacerbated because we usually teach in two dimensions for simplicity—especially in textbooks.1 There are certain rules that have to be followed when displaying a three-dimensional object on a two-dimensional page. This is especially true for metabolites where the stereochemistry is crucial. One of these rules is called the Fischer projection.2
Most students (and faculty) don't understand the relationship between a two-dimensional Fischer projection and the three-dimensional molecule it's supposed to represent. This is unfortunate because it means they don't really understand the three-dimensional conformation of metabolites.
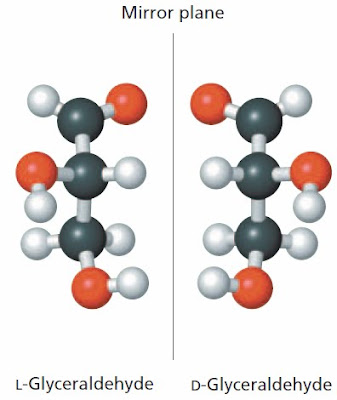
Let's look at a simple three-carbon compound—glyceraldehyde. There are two different versions of glyceraldehyde: D-glyceraldehyde and L-glyceraldehyde. The two different molecules cannot be superimposed, that's why you know that they are different molecules. Enzymes can tell the difference; that's why D-glyceraldehyde is a common metabolite and L-glyceraldehyde is rarely found in cells.
Read more »
No comments:
Post a Comment